Prof. Dr. Sebastian Henke
Technische Universität Dortmund
Fakultät für Chemie und Chemische Biologie
Anorganische Chemie
Otto-Hahn-Str. 6
44227 Dortmund
Room: C2-05-331
E-Mail: sebastian.henke@tu-dortmund.de
Phone: +49 231 755 3976
Fax: +49 231 755 5048

Welcome to our webpage. We are materials chemists working at the interface of solid-state and molecular chemistry. Our goal is to construct functional materials via a modular approach utilising Werner-type coordination chemistry. By self-assembly of inorganic and organic building units we synthesize extended (2D or 3D) coordination networks (or metal-organic frameworks, MOFs) with interesting chemical and physical properties (porosity, flexibility, disorder, etc.). Ultimately, we want to modulate the functional properties of our materials systematically by chemical principles.
Open Positions
Several exciting research topics are available for Bachelor and Master theses:
– Stimuli-responsive MOFs for gas separations and energy storage applications
– MOF glasses for applications as membranes and solid electrolytes
Please contact Sebastian Henke by email if interested.
News
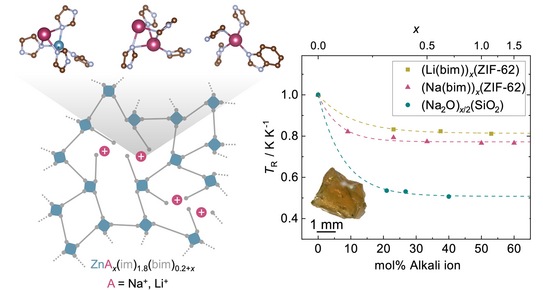
Alkali-Ion Modification of MOF Glasses Published in Nature Chemistry
Pascal’s major PhD project on the chemical modification of metal-organic framework glasses has been published.
P. Kolodzeiski, B. M. Gallant, L. Richter, M. A. T. Ongkiko, C. Franke, A. Kostka, W.-L. Xue, C. Das, J.-B. Weiß, E. Kolodzeiski, T. Kress, G. Kieslich, T. Li, A. J. Morris, D. Kubicki, S. Henke
Alkali-ion-modified zeolitic imidazolate framework glasses
Nat. Chem. 2026, DOI: 10.1038/s41557-026-02115-8
We demonstrate that alkali benzimidazolates can act as network modifiers for MOF glasses, conceptually analogous to modifier oxides in classical silicate glass chemistry. Introducing sodium or lithium benzimidazolates into the melt of the MOF glass former enables systematic tuning of key glass properties, including the glass transition temperature, liquid fragility and density.
Using a combination of X-ray total scattering, atom probe tomography, solid-state NMR spectroscopy and density-functional theory calculations, we reveal how alkali ions integrate into the glass network by partially replacing the original zinc nodes and depolymerising the coordination network. Colleagues from the University of Birmingham made important contributions through advanced solid-state NMR spectroscopy and DFT modelling, which helped elucidate the atomic-scale incorporation of the sodium modifier and their structural consequences for the glass network.
Importantly, the modified glasses retain a substantial fraction of their intrinsic microporosity. Moreover, selective removal of the modifiers by water treatment generates additional mesoporosity, yielding hierarchically porous MOF glasses. These results establish a framework for chemically modifying MOF glasses and provide a new concept for tuning both their processing conditions and pore architecture.
Pascal Kolodzeiski receives Doctoral Thesis Award of the Department of Chemistry and Chemical Biology
Dr Pascal Kolodzeiski has been awarded the Doctoral Thesis Award (Promotionspreis) of the Department of Chemistry and Chemical Biology at TU Dortmund in recognition of his outstanding doctoral thesis entitled “Metal-Organic Frameworks: Concepts from Glass Science and High-Pressure Behavior”. The award was presented during this year’s Tag der Chemie.
The Doctoral Thesis Award is presented annually to two doctoral researchers within the department, one in Chemistry and one in Chemical Biology, in acknowledgement of exceptional scientific achievement. We are delighted that Dr Kolodzeiski’s work has been selected for this distinction. We gratefully acknowledge Bayer AG for sponsoring the award.
Deciphering the guest-free structures of flexible MOFs by 3D electron diffraction
A new publication led by our former postdoc Nassos, in collaboration with colleagues from Stockholm University and Ruhr University Bochum, reports a detailed structural investigation of responsive metal-organic frameworks.
A. Koutsianos, E. Svensson Grape, R. Pallach, J. Keupp, R. Schmid, A. K. Inge, S. Henke
Deciphering the guest-free crystal structures and thermal breathing of the flexible metal-organic frameworks ZIF-7 and ZIF-9
Chem. Sci. 2026, DOI: 10.1039/D5SC08614K
The framework materials ZIF-7 and ZIF-9 are well known for their “breathing” behaviour, in which the pore structure changes cooperatively in response to external stimuli such as gas adsorption or temperature. Using microcrystal three-dimensional electron diffraction (3DED) in combination with powder X-ray diffraction, calorimetry and density functional theory calculations, we resolved the crystal structures of the elusive guest-free phases of both frameworks. The study clarifies a long-standing structural ambiguity in the literature for ZIF-7 and reports the corresponding structure of ZIF-9 for the first time. The results reveal dense, strongly distorted frameworks with minimal pore volume and provide new insight into the thermally induced breathing behaviour of these flexible materials.
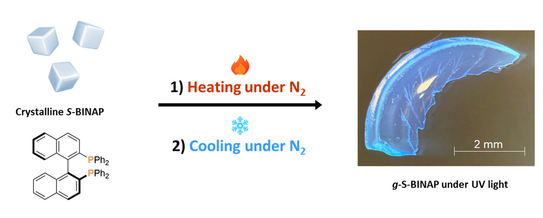
Luminescent Chiral Molecular Glasses from Melt-Quenched BINAP
Nutt’s paper in Advanced Science marks our first collaboration with the Steffen Group.
N. Krittametaporn, P. Ralle, D. Dierks, C. Nelle, S. K. Vasa, P. Kolodzeiski, R. Linser, A. Steffen, S. Henke
Luminescent Chiral Molecular Glasses by Melt-Quenching Enantiopure BINAP
Adv. Sci. 2025, e18879.
In this work, we establish melt-quenching as an effective route to transform the axially chiral molecule BINAP (2,2’-bis(diphenylphosphino)-1,1’-binaphthyl) into stable, optically transparent chiral molecular glasses. While enantiopure R- and S-BINAP readily vitrify upon cooling the melt and exhibit glass transition temperatures near 100 °C, the racemic mixture rac-BINAP rapidly recrystallise. Photophysical measurements performed in the Steffen Group reveal that vitrification induces blue-shifted luminescence relative to the crystalline state and leads to a strong enhancement of circularly polarised luminescence (CPL). The resulting dissymmetry factors reach approximately 10⁻², placing these BINAP glasses among the most active purely organic CPL emitters reported to date.
This study was enabled by close collaborations within the Department of Chemistry and Chemical Biology: in addition to the photophysics contributions from the Steffen Group, solid-state NMR spectroscopy by the Linser Group provided key structural insights into the amorphous materials. Together, these results highlight melt-quenched chiral organic glasses as promising platforms for chiroptical photonic and optoelectronic applications.
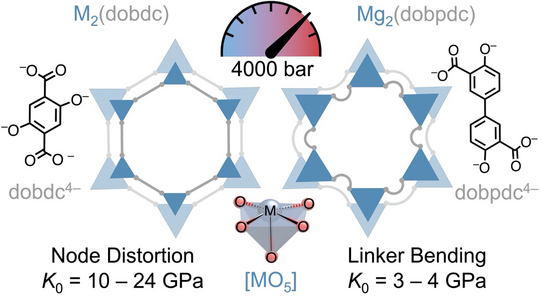
Squeezing MOFs to learn about compression mechanisms and mechanical stability
Pascal’s paper, published in JACS, provides the first comprehensive experimental investigation of the high-pressure behaviour of the important family of MOF-74-type materials.
P. Kolodzeiski, I. de Vries, M. Strobl, L. Frentzel-Beyme, M. Paulus, C. Sternemann, C. Nelle, K. Terlinden, J. Song, N. L. Doltsinis, S. Henke
Understanding the High-Pressure Behavior of Honeycomb-Like Metal-Organic Frameworks with Open Metal Sites: Influence of Metal Identity, Linker Extension and Diamine Functionalization J. Am. Chem. Soc. 2025, 147, 46075.
Using high-pressure powder X-ray diffraction up to 4000 bar, performed at the synchrotron radiation sources DELTA (the TU Dortmund synchrotron) and DESY (Hamburg), the study reveals how metal identity, coordination chemistry, and linker geometry govern compression mechanisms and mechanical stability in MOF-74-type frameworks. The results uncover distinct pressure-response pathways for classical MOF-74 materials and their expanded analogues, and demonstrate how diamine functionalisation can stabilise large-pore frameworks against irreversible pressure-induced collapse. Complementary molecular dynamics simulations, carried out in collaboration with the Doltsinis group (University of Münster), provide atomistic insight into the unusual high-pressure phase behaviour of the diamine-loaded material and rationalise the experimental diffraction data.
Together, these findings establish fundamental structure-property relationships that are highly relevant for the deployment of MOF-74-type materials in pressure-intensive technologies, including carbon capture, catalysis, and membrane fabrication.
Sebastian Henke receives Dalton Horizon Prize for pioneering hybrid glass research
Sebastian has been awarded the Dalton Horizon Prize by the Royal Society of Chemistry, together with 17 international researchers, for their joint discovery and characterisation of hybrid glasses – a new, fourth class of glass materials.
Led by Tom Bennett (University of Canterbury, New Zealand), the team established metal-organic framework glasses as a distinct class alongside inorganic, organic, and metallic glasses. The contributions of the Henke Group focused on understanding the porosity, magnetism, and chemistry of these novel materials, revealing key structure-property relationships relevant for gas separation, sensing, and optical applications.
This was a truly international team effort that brought together complementary expertise from across the globe. We are proud to have contributed to shaping the fundamental understanding of this exciting new family of glasses.
Iron-Based MOF Glass Exhibits Frustrated Cooperative Magnetism
In our latest Advanced Functional Materials paper, we unveil low-temperature dynamic magnetism in a new iron(II)-based MOF glass.
C. Das, S. Salamon, J. Landers, J.-B. Weiß, W.-L. Xue, P. Kolodzeiski, R. Pallach, H. Wende, S. Henke
Emergent Spin-Glass Behavior in an Iron(II)-Based Metal-Organic Framework Glass
Adv. Funct. Mater. 2025, DOI: 10.1002/adfm.202517854
In this study, led by former postdoctoral researcher Chinmoy Das, we present a solvent-free, one-pot synthesis of an iron(II)-based MOF glass that exhibits spin-glass freezing around 14 K. Despite the absence of long-range structural and topological order, its amorphous network gives rise to frustrated, short-range magnetic exchange interactions, resulting in cooperative magnetic behaviour at low temperatures. This finding demonstrates that amorphous hybrid materials can exhibit rich collective magnetic phenomena, thereby extending the design space of functional materials beyond crystalline frameworks. We gratefully acknowledge our collaborators Soma Salamon, Joachim Landers, and Heiko Wende (University of Duisburg-Essen) for their key contributions to the magnetic characterisation.
Tim Krokowski received a Poster Prize at the MOFschool 2025 at Lake Como
We are delighted that our PhD student Tim Krokowski was awarded a Best Poster Presentation Prize at the 4th International School on Porous Materials: MOFschool2025, held from 16–20 June 2025 at Lake Como, Italy.Tim presented his research entitled “Reticular Design of Soluble Porous Salt Frameworks”, showcasing an advanced and modified generation of porous salt frameworks (PSFs) – a class of soluble porous materials recently developed by our group (see publication). The MOF-based materials are distinguished by their solubility and versatile processability
A key innovation of PSFs is the use of Na⁺ ions as inorganic building blocks. In contrast to conventional MOFs, which typically rely on transition metal cations that are often scarce or costly, sodium is a major component of common salt and is readily available in large quantities at a low cost. This aspect significantly enhances the sustainability and scalability of the material class. Tim’s work focuses on developing environmentally friendly synthesis methods and introducing precise structural modifications to obtain more robust frameworks with larger pore diameters. These features open new possibilities for the use of PSFs in applications such as the selective separation of gas and vapour mixtures.
The award was generously sponsored by Frontiers in Chemistry. Congratulations, Tim!
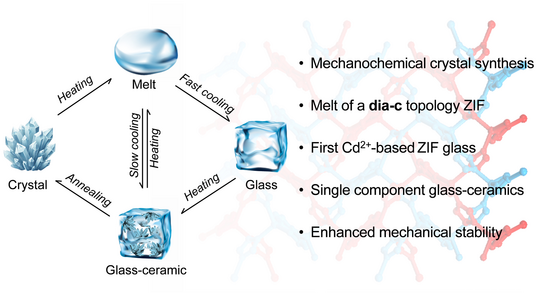
MOF glass-ceramics
Wen-Long’s latest paper, reporting the formation of MOF glasses and glass-ceramics from a cadmium-based zeolitic imidazolate framework, has been published in JACS.
W.-L. Xue, A. Klein, M. El Skafi, J.-B. Weiß, F. Egger, H. Ding, S. K. Vasa, C. Liebscher, M. Zobel, R. Linser, J.-C. Tan, S. Henke,
Mechanochemical Synthesis Enables Melting, Glass Formation and Glass–Ceramic Conversion in a Cadmium-Based Zeolitic Imidazolate Framework,
J. Am. Chem. Soc. 2025, 147, 15625.
Metal-organic framework (MOF) glasses are typically produced by melt-quenching first-row transition metal ZIFs. In this study, Wen-Long shows that mechanochemical synthesis can enable melting and glass formation of a cadmium-based ZIF (cadmium imidazolate, Cd(im)₂) by reducing its crystallite size and increasing defect density, thereby lowering its melting point below its decomposition temperature. Rapid quenching yields a stable monolithic glass, while controlled reheating produces a single-component glass-ceramic with nanocrystalline domains embedded in the glassy matrix. This approach not only extends meltable MOF systems beyond first-row transition metals but also demonstrates the first example of a MOF-derived glass-ceramic with enhanced mechanical properties.
Best Presentation Award for Jan-Benedikt Weiß
At this year’s Tag der Chemie (Chemistry Day) of our department, Jan-Benedikt Weiß received a best presentation award for his outstanding talk entitled Novel Hybrid Glasses via Ligand Exchange and Addition Reactions in Melts. In his presentation, Jan-Benedikt introduced a groundbreaking new strategy for the targeted modulation of the structure and network topology of MOF glasses. The jury was particularly impressed by the scientific depth, conceptual clarity, and originality of his work.
Congratulations to Jan-Benedikt on this well-deserved recognition — and stay tuned for the upcoming publication!
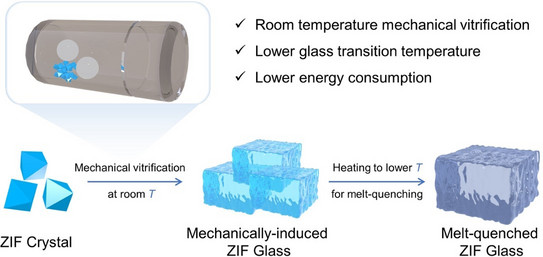
Creating Porous Glasses at Room Temperature Through Mechanochemistry
Wen-Long’s second paper from his PhD studies, which demonstrates the preparation of MOF glasses through mechanochemistry, was published in Angewandte Chemie.
Insights into the Mechanochemical Glass Formation of Zeolitic Imidazolate Frameworks
W.-L. Xue, C. Das, J.-B. Weiß, S. Henke
Angew. Chem. Int. Ed. 2024, 63, e202405307
Metal-organic framework (MOF) glasses, known for their potential in gas separation, optics, and solid-state electrolytes, benefit from the processability of their supercooled liquid state. Traditionally, MOF glasses are produced by heating MOF crystals to their melting point and then cooling the liquid to room temperature under an inert atmosphere. While effective, this melt-quenching technique is energy-intensive due to the high temperatures required. Additionally, it limits the development of new materials to combinations of metal ions and linkers that are highly thermally stable.
Wen-Long has demonstrated that some crystalline zeolitic imidazolate frameworks (ZIFs), a subset of MOFs, can be transformed into glasses in just 5 minutes by mechanical milling at room temperature. This milling method can also transform crystalline ZIFs in the glassy state that do not melt due to low thermal stability. Notably, this technique enabled the preparation of the first thermally sensitive Cu-based ZIF glass via mechanochemistry.
Creating MOF glasses through mechanical milling at room temperature is not only more energy-efficient but also allows for the incorporation of thermally sensitive materials, such as organic or organometallic molecules, into versatile MOF glass composites. This approach broadens the spectrum of potential applications, paving the way for the development of multifunctional MOF glass composites and expanding their use into more diverse fields.
Jan-Benedikt Weiß Embarks on Research Collaboration at Kyoto University
Our PhD student, Jan-Benedikt Weiß, has commenced a six-month research visit to the esteemed laboratory of Professor Satoshi Horike at Kyoto University. Professor Horike is renowned globally for his pioneering work in the field of metal-organic framework (MOF) glasses, particularly for their applications in solid-state ionics, catalysis, and sorption.
During his stay, Jan will focus on exploring innovative methods for the organic modification of MOF glasses. His research aims to design and characterize novel MOF glasses that exhibit metal-centered redox activity, potentially leading to significant advancements in this cutting-edge area of materials chemistry. We extend our gratitude to Professor Horike and his team for their generous support, access to their world-class research facilities, and for welcoming Jan to the rich cultural experience of life in Japan.
Highly Porous MOF Glasses by Compositional Engineering
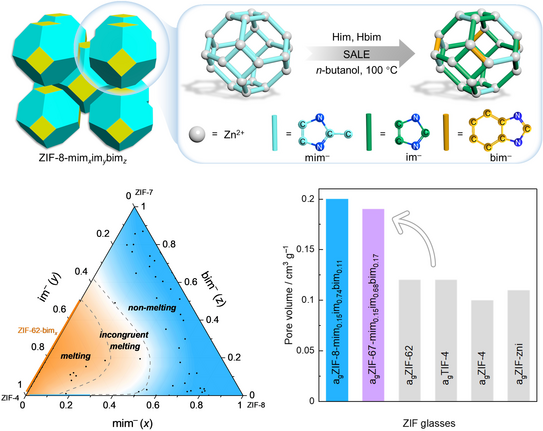
Wenlong’s paper on using a ligand exchange strategy to prepare highly porous MOF glasses was published in Nature Communications.
Highly porous metal-organic framework liquids and glasses via a solvent-assisted linker exchange strategy of ZIF-8
W.-L. Xue, P. Kolodzeiski, H. Aucharova, S. Vasa, A. Koutsianos, R. Pallach, J. Song, L. Frentzel-Beyme, R. Linser, S. Henke
Nat. Commun. 2024, 15, 4420.
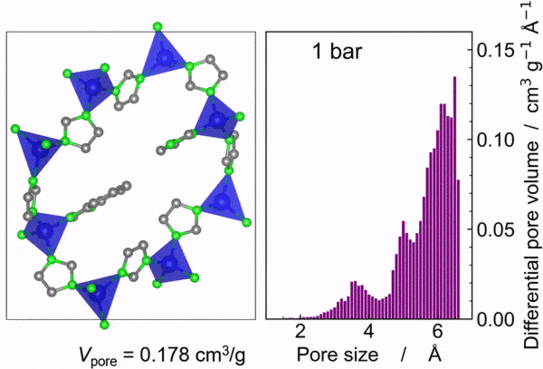
Wen-Long has successfully utilised solvent-assisted linker exchange (SALE) to modify a highly porous metal-organic framework (MOF) by partially replacing its organic linkers with ones that possess weaker metal-linker bond strengths. This alteration enables the MOF derivatives to transition through crystal, liquid, and glass phases upon thermal treatment. Wen-Long produced a comprehensive series of 50 modified MOF derivatives and conducted detailed thermal and crystallographic analyses. These efforts led to the construction of a ternary phase diagram that delineates the compositional thresholds necessary for melting and glass formation. Notably, the resulting MOF glasses demonstrated a much-increased specific pore volume (approximately 0.20 cm3/g) compared to previous MOF glasses. Additionally, gas physisorption tests revealed that these glasses can adsorb substantial amounts of technologically relevant C3 and C4 hydrocarbons and can effectively separate propylene from propane based on their adsorption kinetics.
The limited number of meltable MOFs known to date underscores the potential of Wen-Long's linker exchange strategy, opening new pathways for creating meltable and glass-forming MOFs from the extensive pool of non-meltable MOFs documented in the literature. This breakthrough not only advances the science of MOF glasses but also paves the way for future investigations into the development of highly porous, moldable materials for diverse technological uses.
Lennard Richter visits the University of Oxford
Lennard Richter is undertaking a three-month internship at the Multifunctional Materials & Composites (MMC) Laboratory at the Department of Engineering Science of the University of Oxford. The internship is part of his Master´s thesis in which he investigates new routes for the chemical modification of metal-organic framework glasses. Lennard will investigate the mechanical properties of these novel materials using nanoindentation and atomic force microscopy. He also aims to gain new insights into the glasses’ chemical structure via spatially resolved nano-FTIR spectroscopy. We are very grateful to Prof. Jin-Chong Tan and his group for sharing their expertise in these methods. The Heinrich J. Klein Foundation, administered by SCHOTT, is acknowledged for generously sponsoring this exchange with a fellowship for Lennard.
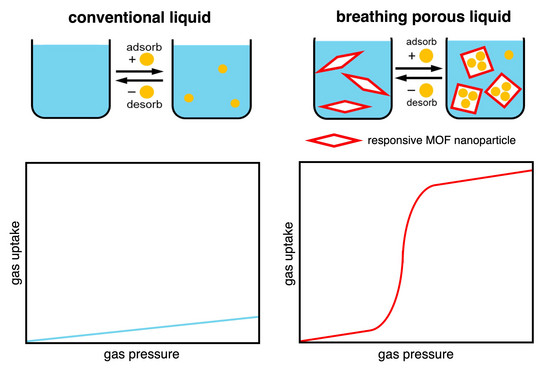
Breathing Liquids
Nassos’ paper about breathing porous liquids based on submicron-sized responsive MOF particles was published in Nature Communications.
Breathing porous liquids based on responsive metal-organic framework particles
A. Koutsianos, R. Pallach, L. Frentzel-Beyme, C. Das, M. Paulus, C. Sternemann, S. Henke
Nat. Commun. 2023, 14, 4200.
According to Henry’s law, typical liquids show a linear increase of gas solubility with increasing gas pressure. Nature, however, provides an impressive example of a liquid with a step-shaped gas saturation curve. Owing to a cooperative structural transformation of haemoglobin upon oxygen binding, the oxygen saturation curve of blood displays a characteristic sigmoidal shape as a function of the oxygen partial pressure. The sigmoidal shape of the oxygen saturation curve of blood is of the highest relevance for an efficient oxygen transport mechanism from the respiratory organs to the tissues and thus a key factor for metabolism in almost all vertebrates.
The colloidal liquid systems developed by Nassos, exhibit a breathing behaviour characterized by sigmoidal gas saturation curves, similar to the distinct oxygen saturation curve of blood. We report that dispersions of responsive MOF particles in a size-excluded silicone oil form stable porous liquids exhibiting gated uptake for CO2, propane, and propylene, as characterized by sigmoidal gas sorption isotherms with distinct transition steps. Due to the sigmoidal gas saturation curve, most of the gases adsorbed in the liquid system can be released by a relatively small change in gas pressure or temperature, making these systems potentially more energetically efficient than conventional liquid adsorbents. With the help of in situ X-ray diffraction experiments, Nassos could show that the characteristic gas saturation curve of the MOF dispersions is caused by a narrow pore to large pore phase transformation of the responsive MOF particles, which react to gas pressure despite being dispersed in silicone oil. Given the established flexible nature and tunability of a range of MOFs, these results herald the advent of breathing porous liquids whose sorption properties can be tuned rationally for a variety of technological applications.
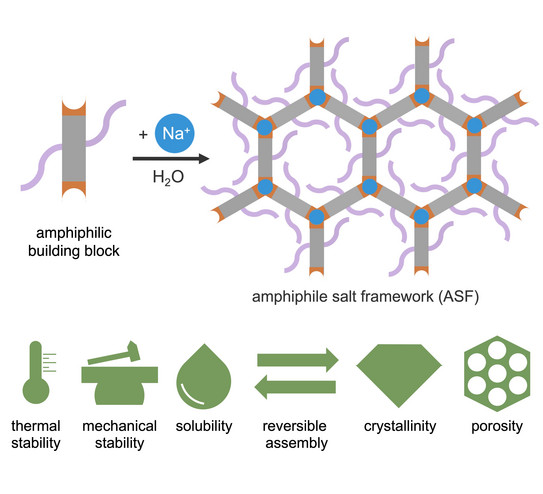
Porous Soap
Louis Frentzel-Beyme’s paper reporting a new class of water-processible porous materials was published in Advanced Functional Materials.
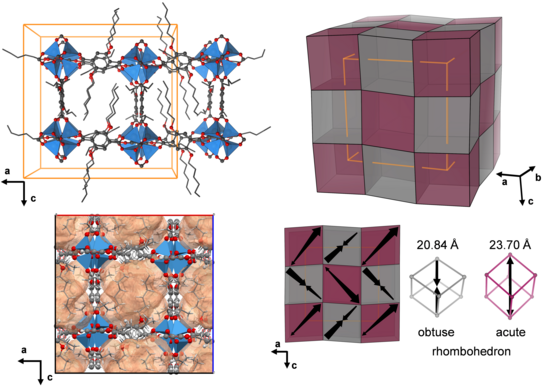
Microporous, Crystalline, and Water-Processable Framework Materials of Organic Amphiphile Salts
L. Frentzel-Beyme, P. Kolodzeiski, K. Terlinden, S. Henke
Adv. Funct. Mater. 2023, 33, 2302033.
Porous materials are indispensable for a variety of technological processes. Their most important areas of application are found in molecular separation, purification, sensor technology and catalysis. In this paper we present a novel class of crystalline porous materials consisting of amphiphilic organic molecules and sodium ions. Chemically, these amphiphilic salt frameworks (ASFs) are closely related to conventional soaps; they are sodium salts of organic acids with two lipophilic (fat-loving) carbon chains and two hydrophilic (water-loving) head groups. The special feature of ASFs is that they dissolve in water and break down into their molecular building blocks, while they reassemble into the highly ordered porous salt frameworks when the water evaporates. Because of this property, which is unique for porous materials, the ASFs can be processed in aqueous solution and easily regenerated. In addition, the crystalline materials can also be synthesised mechanochemically by intensive grinding of the organic acid molecules with sodium hydroxide in the absence of any solvent. With elaborate gas sorption experiments, we show that the porous soaps can store various technologically relevant gases, such as carbon dioxide or short-chain hydrocarbons, in their pores and even possess potential for molecular separation. X-ray diffraction experiments, in which the materials were subjected to extremely high pressures of up to 4000 bar (i.e. 4000 times the atmospheric pressure), also attest the ASFs' excellent mechanical stability. The high-pressure experiments were conducted at the Dortmund synchrotron DELTA.
New Paper on Liquid and Glassy MOFs published in JACS
Jianbo Song’s paper dealing with cyano-functionalized crystalline MOFs, their liquids and glasses was published in the Journal of the American Chemical Society.
Modulating Liquid–Liquid Transitions and Glass Formation in Zeolitic Imidazolate Frameworks by Decoration with Electron-Withdrawing Cyano Groups
J. Song, L. Frentzel-Beyme, R. Pallach, P. Kolodzeiski, A. Koutsianos, W.-L. Xue, R. Schmid, S. Henke
J. Am. Chem. Soc. 2023, 145, 9273
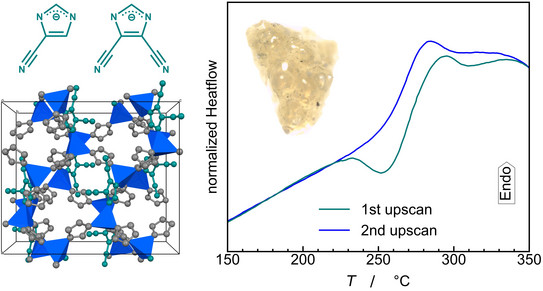
We report that the implementation of imidazolate-type linkers featuring strongly electron-withdrawing cyano groups substantially lowers the melting point of zeolitic imidazolate frameworks (ZIFs). By quenching the liquid ZIFs to room temperature porous glasses with surprisingly low glass transition temperatures are obtained. Detailed thermal analyses with differential scanning calorimetry (DSC) provide deep insights into the thermodynamics of framework collapse, melting and glass formation of this extraordinary materials. Excitingly, the calorimetric data suggest that the cyano-functionalized ZIFs possess polyamorphism, i.e. they form two distinct liquid phases with different porosity, density, enthalpy and entropy. The results form the basis for the development of highly porous MOF glasses with low glass transition temperatures and MOF liquids with comparatively low viscosity.
Awards for Kai Terlinden and Jan-Benedikt Weiß
At the Tag der Chemie (Chemistry Day) of our department two of our group members were honoured. Kai Terlinden received one of the poster prizes for his outstanding poster on the immobilisation of luminescent metal complexes in MOFs. Jan-Benedikt Weiß was award a price for the best Master of Chemistry degree of the last academic year. Congratulations to you both for these well-deserved recognitions!
Insights into temperature-driven disorder-order transitions of responsive MOFs
Roman Pallach’s paper on the intriguing structural response of his aperiodically distorted MOFs has been published in the journal APL Materials. The paper is part of a special issue to celebrate the 75th birthday of Professor Sir Anthony K. Cheetham, the postdoc advisor of Sebastian Henke.
Entropy Driven Disorder-Order Transition of a Metal-Organic Framework with Frustrated Flexibility
Roman Pallach, Jan-Benedikt Weiß, Katrin Vollmari, Sebastian Henke
APL Mater. 2023, 11, 041118
The unprecedented disorder-order phase behaviour of his alkoxy-functionalized MOFs was previously reported by Roman in Nature Communications. The atomistic mechanism of the phase change, however, was so far unclear. In contrast to ordinary materials’ behaviour, the long-range order in the functionalized MOF increases when the temperature rises. With the help of X-ray scattering, X-ray diffraction and infrared spectroscopy, all recorded at variable temperature in the range from 25 °C to about 225 °C, Roman was now able decipher the mechanism of the disorder-order transition, by which an apeoridically distorted framework relaxes toward a periodic arrangement. The driving force for the ordering of the framework backbone at high temperature is increased “liquid-like” disorder of the alkoxy side chains pinned to the organic linkers, i.e. their increased configurational and vibrational entropy. The experimental findings were supported by DFT calculations performed by Jan-Benedikt Weiß. The results are of relevance for the development of novel responsive materials with switchable optical or catalytic properties.
Highlighted on the Cover Image of the Issue April 2023 of APL Materials.
Another doctor from the group
Jianbo Song successfully defended his PhD thesis on 20th March 2023. Many congratulations! He was the third PhD student to join our group in the late summer 2019. His outstanding results have been compiled in the thesis entitled “Functionalized Zeolitic Imidazolate Frameworks – Tuning the Responsive Phase Transition and Melting Behavior”. Jianbo worked on two exciting topics:
- The structural response of flexible MOFs of the ZIF-62 family under mechanical pressure up to 4000 bar (published in Angewandte Chemie)
- The melting behaviour of cyano-functionalized ZIFs and the formation of porous MOF glasses thereof (publication coming soon, stay tuned)
Kekulé Fellowship for Jan-Benedikt Weiß
Jan-Benedikt Weiß receives a prestigious Kekulé Fellowship of the "Fonds der Chemischen Industrie" (FCI) to support his doctoral research project on MOF glasses in our group. We are very proud that Jan passed the rigorous selection process and thank the FCI for its generous funding. Building on the results of his outstanding Master's thesis in 2022, Jan is now looking into designing and understanding new glassy MOF materials with tailored porosity and additional metal-centred functionality (i.e. redox activity). The results may open new doors for the applications of MOF glasses in thermoelectrics, optoelectronics and membranes.
Jan-Benedikt Weiß receives a poster prize at the 11th Young Chemists Symposium Ruhr 2023
Under the theme "Creating Sustainability", the 11th Young Chemists Symposium Ruhr (JungChemiker Symposium Ruhr) took place at TU Dortmund University. We are delighted that our doctoral student Jan-Benedikt Weiß received one of the two poster prizes of the symposium for showcasing his exciting work on new glassy materials based on cobalt imidazolate frameworks. Many congratulations!
New paper finally out in Nature Communications
Louis’ paper on the porosity in metal-organic framework (MOF) glasses is now published in Nature Communications.
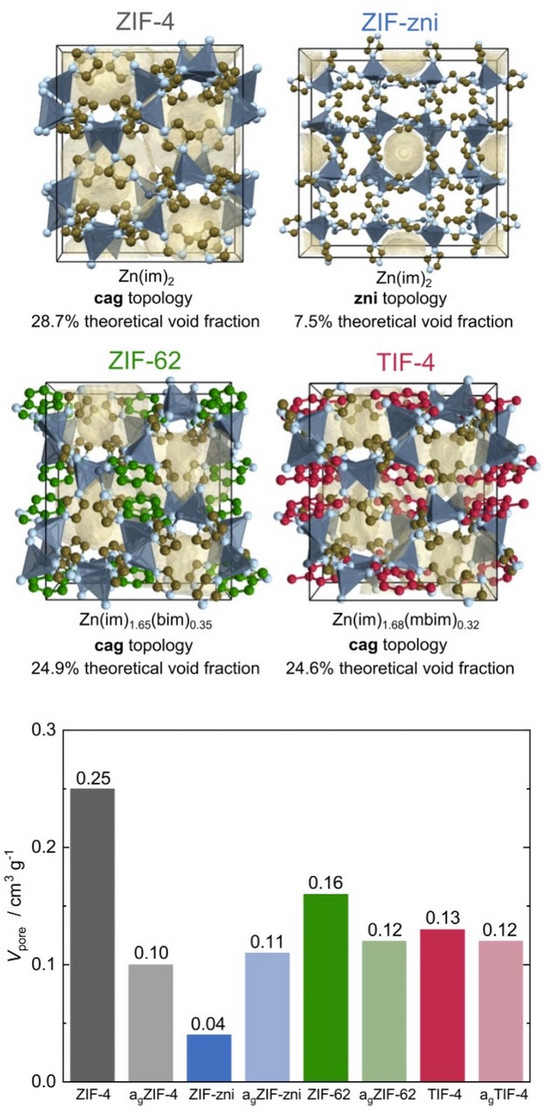
Quantification of gas-accessible microporosity in metal-organic framework glasses
L. Frentzel-Beyme, P. Kolodzeiski, J.-B. Weiß, A. Schneemann, S. Henke
Nat. Commun. 2022, 13, 7750
MOF glasses are a new class of glass materials; the first example was reported back in 2015. These glasses are obtained by heating a crystalline MOF to a high temperature until a melt forms. On cooling, the melt vitrifies and a MOF glass is formed, i.e. a disordered solid with the structure of a "frozen" liquid. The liquid phase of MOFs opens up new avenues for shaping and processing the material. Since the crystalline MOF glass precursors are microporous materials, it was assumed that the corresponding glasses would have similar properties. However, gas-accessible microporosity in MOF glasses has been poorly studied thus far. This work represents the first comprehensive study of microporosity in MOF glasses and also provides valuable design principles for the development of new porous MOF glasses with tailored properties. The gained understanding is of importance for future applications of MOF glasses in various fields, such as gas separation membranes or solid electrolytes.
Roman Pallach defended his PhD thesis
Roman Pallach successfully defended his dissertation on 17 October 2022 – a second milestone for the Henke Group, not only because of the fancy doctor's hat with integrated wig and built-in guitar cable. Roman is a founding member of the Henke Group and played a key role in setting up the labs in the early days. His dissertation, entitled "The Impact of Alkyl- and Alkoxy- functionalization on the Responsive Behaviour of Metal-Organic Frameworks: From Conventional to Frustrated Flexibility", describes how the responsiveness of MOFs can be controlled by decorating the organic linkers with flexible alkyl or alkoxy groups. His most important achievement was the development of the concept of frustrated flexibility, which was published in Nature Communications in 2021 (https://doi.org/10.1038/s41467-021-24188-4). Further publications by Roman on responsive MOFs with unusual behaviour will follow soon.
The Henke Group participated in the international conference MOF2022 in Dresden
From 4th to 7th September, (almost) the entire Henke Group attended the 8th International Conference on Metal-Organic Frameworks and Open Framework Compounds (MOF2022) in the beautiful city of Dresden. The group was represented with nine posters from the PhD students and postdocs as well as a keynote by Sebastian. It was great to exchange ideas and initiate new collaborations with so many other researchers in the field.
Jan-Benedikt Weiß visits Oxford University for three months

As part of the research for his Master thesis, Jan-Benedikt Weiß is visiting the world-renowned group of Prof. Andrew Goodwin at the Inorganic Chemistry Laboratory of the University of Oxford. For three months, Jan learns new skills for modelling disordered and glassy framework materials based on X-ray total scattering data. While at Oxford, Jan will also visit the synchrotron radiation facility Diamond Light Source to collect high-quality X-ray total scattering data from his glassy MOF samples. We are very grateful to Andrew Goodwin and his group for their outstanding hospitality. Jan would also like to thank SCHOTT for sponsoring this project with a scholarship from the Heinrich J. Klein Foundation.
First PhD from the group completed
Louis Frentzel-Beyme successfully defended his doctoral thesis. This is a milestone for the Henke group, as it is the group's first doctoral degree. Louis' dissertation, entitled "Towards Processable Metal-Organic Frameworks - Porous Glasses and Water-Processable Materials," provides new insights, particularly in the emerging field of MOF glasses. One of his breakthroughs is the development of a linker exchange strategy that allows systematic tuning of the melting point of MOF crystals as well as the glass transition point of the corresponding glasses.
In addition, Louis has gained fundamental insights into the porosity of MOF glasses and demonstrated their potential for gas separation applications. Check out Louis' publications for a deeper dive into the subject:
L. Frentzel-Beyme, M. Kloß, R. Pallach, S. Salamon, H. Moldenhauer, J. Landers, H. Wende, J. Debus, S. Henke
J. Mater. Chem. A 2019, 7, 985
DOI: 10.1039/C8TA08016J
L. Frentzel-Beyme, M. Kloss, P. Kolodzeiski, R. Pallach, S. Henke
J. Am. Chem. Soc. 2019, 141, 12362
DOI: 10.1021/jacs.9b05558
L. Frentzel-Beyme, P. Kolodzeiski, J.-B. Weiß, S. Henke
Preprint on ChemRxiv, 2022
DOI: 10.26434/chemrxiv-2021-lq308
Fine-tuning the mechanics of responsive MOFs
Jianbo’s paper on the high-pressure phase behaviour of flexible zeolitic imidazolate frameworks (ZIFs) has just been published in Angewandte Chemie.
Tuning the High-Pressure Phase Behaviour of Highly Compressible Zeolitic Imidazolate Frameworks: From Discontinuous to Continuous Pore Closure by Linker Substitution
J. Song, R. Pallach, L. Frentzel-Beyme, P. Kolodzeiski, G. Kieslich, P. Vervoorts, C. L. Hobday, S. Henke, Angew. Chem. Int. Ed. 2022, e202117565
Sophisticated high-pressure powder X-ray diffraction experiments, which have been performed at the synchrotron radiation source Diamond Light Source (UK), shed light on the unusual high-pressure response of a series of porous ZIFs. The open framework structures reversibly transform to a closed framework under application of mechanical pressure. Interestingly, the pressure driven phase transformation can be switched from discontinuous to continuous by partially substituting the small imidazolate building units by larger benzimidazolate units. The continuous transition from the fully open to the closed structure allows precise modulation of the materials pore size by application of mechanical pressure to the material. This finding is of relevance for the utilization of such flexible framework materials in gas separation membranes, shock absorbers and nanoscopic actuators.
Gateway Fellowship for Louis Frentzel-Beyme

Louis has been awarded with the Gateway Fellowship for post-doctoral researchers of the COORNETs DFG Priority Program 1928. During the one-year financial support, Louis will continue his research in the emerging field of MOF glasses. Here, he aims to develop new functionalized MOF glass composites for potential applications in e.g. gas separation or optics. The integration in the interdisciplinary COORNETs program, which includes many renowed research groups in Germany, is an excellent opportunity to take the first steps towards an independent scientific career. Congratulations and we are happy to have you in the group for another year!
Sebastian Henke receives TU Dortmund Teaching Price 2021 together with Prof. Andreas Steffen
Sebastian Henke and Andreas Steffen have been awarded the Teaching Price of TU Dortmund University in the category 1 (courses with more than 60 participants) for their great commitment to the first-semester lecture "General and Inorganic Chemistry (AC1)" in winter semester 2020/21. Please see the website of the Annual Academic Celebration (Akademische Jahresfeier) of TU Dortmund University for more details (in German).
Squeezing responsive MOFs under high pressure
In collaboration with the group of Prof. Stephen Moggach from the University of Western Australia (Perth) and an international team we studied how guest-responsive MOFs behave when exposed to mechanical pressures up to 2.1 GPa (that is approx. 21,000 times the atmospheric pressure). The study has been published in the journal Chemical Science.
Guest-Mediated Phase Transitions in a Flexible Pillared-Layered Metal-Organic Framework under High-Pressure
G. Turner, S. C. McKellar, D. R. Allan, A. K. Cheetham, S. Henke*, S. A. Moggach*
Chem. Sci. 2021, 12, 13793-13801
We performed single crystal X-ray diffraction experiments on a responsive MOF using a diamond anvil cell for the high pressure environment. The MOF shows a drastically different mechanical phase behaviour and distinct network distortions depending on the type of guest molecule in its pores. Our results demonstrate the large influence of guest molecules on the high-pressure phase behavior of responsive MOFs. Guest-mediated framework flexibility is useful to engineering MOFs with bespoke pore shapes and compressibility.
New paper published in Nature Communications: Frustrated flexibility in metal-organic frameworks
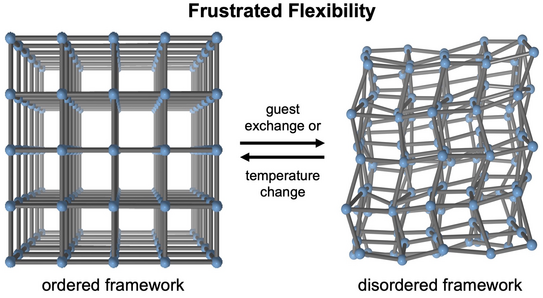
Roman's paper on the unusual structural responsiveness of alkoxy-functionalized MOF-5 derivatives is now published in Nature Communications.
Frustrated flexibility in metal-organic frameworks
R. Pallach, J. Keupp, K. Terlinden, L. Frentzel-Beyme, M. Kloß, A. Machalica, J. Kotschy, S. K. Vasa, P. A. Chater, C. Sternemann, M. T. Wharmby, R. Linser, R. Schmid, S. Henke
Nat. Commun. 2021, 12, 4097
We report a strategy to create MOFs with a ‘frustrated’ structure arising from an incompatibility of intra-framework dispersion forces with the geometrical constraints of the framework’s inorganic building units. Mediated by guest exchange or temperature changes, the frustrated MOFs undergo reversible loss and recovery of crystalline order while preserving framework connectivity and topology. Some of these frustrated MOFs own unprecedented physical properties, such as continuous non-crystalline-to-crystalline transitions driven by entropy rather than enthalpy. The novel phenomenon of frustrated flexibility has consequences for the application of MOFs in gas storage, separation, and catalysis, and further suggests great potential for the discovery of new responsive materials exhibiting unconventional and exotic properties.
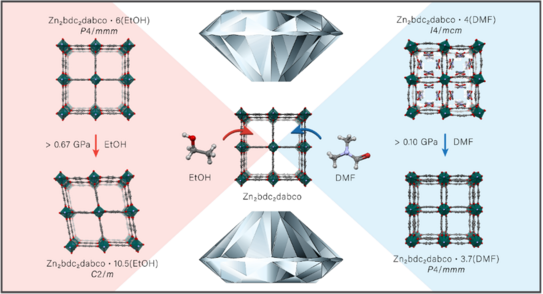
Awards for Kai Terlinden and Pascal Kolodzeiski
At the (virtual) 14th Day of Chemistry the best graduates of the Department CCB of the past year were honoured. Congratulations to our students Kai Terlinden and Pascal Kolodzeiski for receiving the award for their outstanding master's degrees. In his Master's thesis, Kai developed an isoreticular series of novel alkali ion based porous framework compounds, which can be processed from ethanolic solution. Pascal used sophisticated in situ X-ray diffraction and scattering techniques to look into the mechanical and thermal behaviour of various prototypical MOFs. We are very pleased that Kai and Pascal stay in the group for their doctorate.
Sebastian Henke appointed to the rank of Professor
Sebastian has been appointed to a new professorship in the field of Inorganic Chemistry. With this new role the Henke Group will continue to grow and to tackle eminent questions in the materials chemistry of coordination networks and metal-organic frameworks (MOFs). This success would not have been possible without the hard and dedicated work of our excellent PhD, Master and Bachelor students, as well as the outstanding postdocs, who have been working in our group over the past four years. A big “Thank You!” goes to all current and former group members and all colleagues in the Department CCB!
Kekulé Fellowship for Pascal Kolodzeiski

Pascal Kolodzeiski receives a prestigious Kekulé fellowship of the "Fonds der Chemischen Industrie" (FCI) to support his doctoral research project on MOF glasses in our group. We are very proud that Pascal passed the rigorous selection process and thank the FCI for its generous funding. Building on his outstanding Master's thesis in 2020 (see below), Pascal is now looking into designing new functional MOF glasses for applications as solid electrolytes and membrane materials.
Pascal Kolodzeiski receives the "best of the year award" of the Department CCB

Big congratulations to Pascal, who received the best of year award of the Department CCB for his excellent master’s degree in 2020. In his master’s thesis, entitled "Investigations on the structural behaviour of carboxylate- and imidazolate-based metal-organic frameworks under mechanical pressure and high temperature", Pascal studied the mechanical and thermal response of a number of prototypical MOFs with in situ X-ray diffraction and scattering techniques. Parts of this work were performed at DELTA, the synchrotron radiation facility of TU Dortmund. We are very happy that Pascal decided to stay with us and study towards a PhD in our lab.
Exploration Grant of the Boehringer Ingelheim Foundation to investigate the ionic conductivity of MOF glasses

The Boehringer Ingelheim Foundation funds our project to study the ionic conductivity of MOF glasses. MOF glasses are a new class of nanoporous solids that can be processed in their liquid state, which is a conceptual advantage over the classical crystalline solid electrolytes. For this project we are looking for a talented electrochemist with profound experience in electrochemical impedance spectroscopy (postdoc level) to join our group. Please see the job ad. Applications of interested candidates can be send by email to Sebastian Henke.
Experiment! – New grant from the Volkswagen Foundation to explore porous liquids
The Volkswagen Foundation approved our grant in the frame of the funding program "Experiment!” In this explorative project, we will investigate the gas sorption behaviour of porous liquids based on colloidal metal-organic framework (MOF) suspensions. For this purpose, we are seeking a highly motivated and talented postdoc to join our group. If you are interested, please see the job ad.
Chiral Glow - DFG funds our joint research project with the Steffen Group
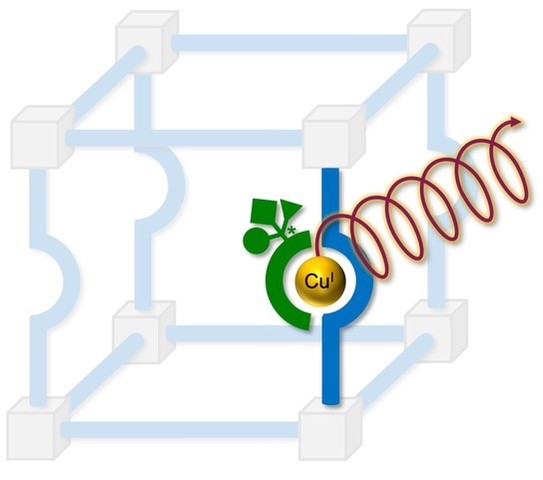
Within the frame of the DFG priority programme 1928 COORNETs (Coordination Networks: Building Blocks for Functional Systems) we received funding for an exciting collaborative project with the group of Prof. Andreas Steffen. The Steffen and Henke Groups will join forces to utilize specifically designed MOFs as host matrices for chiral organometallic Cu(I) complexes. These chiral compounds can exhibit circularly polarized luminescence (CPL) with a yet to fully explore potential in enantioselective sensors, data storage, (3D-)OLEDs, or ultrafast switching in quantum cryptographical applications. We will follow a specific design strategy to obtain beneficial CPL properties in single crystals, powders and films, and finally employ these new materials in CP-PhOLEDs (circularly polarized phosphorescent organic light-emitting diodes).
EuroMOF 2019 in Paris

Louis, Pascal and Roman presented their work on porous sodium organic salts, flexible frameworks and MOF glasses at "EuroMOF 2019 - The 3rd International Conference on Metal Organic Frameworks and Porous Polymers" in Paris. We thank the DFG priority programme 1928 "COORNETs" and the Gesellschaft Deutscher Chemiker e.V. for their generous support.
Paper Published in JACS: Porous Metal Imidazolate Glasses Can Separate Hydrocarbons
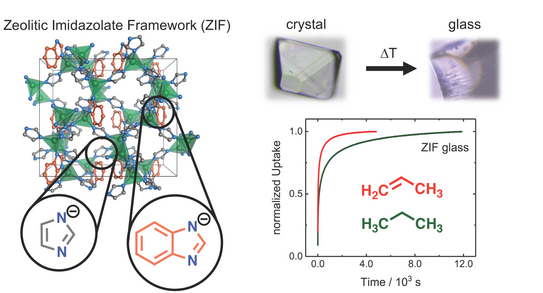
Louis’ paper on zeolitic imidazolate framework (ZIF) glasses has just been accepted for publication by JACS.
Meltable Mixed-Linker Zeolitic Imidazolate Frameworks and Their Microporous Glasses - From Melting Point Engineering to Selective Hydrocarbon Sorption
L. Frentzel-Beyme, M. Kloss, P. Kolodzeiski, R. Pallach, S. Henke*
J. Am. Chem. Soc. 2019, DOI: 10.1021/jacs.9b05558.
We report a synthetic strategy for melting point engineering of crystalline ZIFs. Via a linker mixing approach the melting point of a prototypical ZIF material is decreased to only about 370 °C – a record low for these kind of materials. This sets the stage for the development of lower temperature processing techniques for porous ZIF liquids and glasses. Melting the ZIF crystals followed by cooling the liquid to room temperature yields porous ZIF glasses, which feature pores large enough to adsorb various hydrocarbon gases. Importantly, kinetic sorption profiles indicate that the glasses are able to separate propylene from propane; one of the most important separation problems of the chemical industry.
Spotlight on Elastic Porous Crystals

We are very happy that our Chemical Science Paper on the mechanical-pressure-driven open pore to closed pore phase transitions of a family of zeolitic imidazolate frameworks (ZIFs) has been highlighted in the Annual Review of Diamond Light Source. This work benefited a lot from the world class X-ray diffraction equipment available at beamline I15 of Diamond Light Source.
(see page 60 for our work)
Three Great Theses in 2018

Julia Kuhnt, Marvin Kloß and Stefan Koop performed their Master‘s research projects in our group and successfully defended their theses in 2018. Topics covered range from metal-organic framework glasses and photo-switchable MOFs to hybrid inorganic-organic perovskites. Congratulations and all the best for your future research projects.
Purple, Paramagnetic, Porous - The First Cobalt Imidazolate Glass
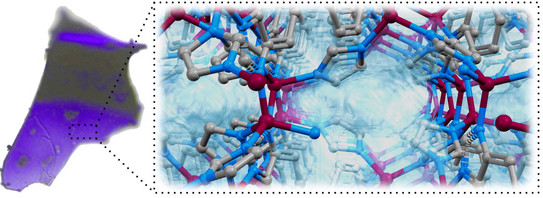
Louis' and Marvin's paper on a permanently porous cobalt-based zeolitic imidazolate framework glass has just been accepted for publication in the Journal of Materials Chemistry A.
"Porous purple glass - A cobalt imidazolate glass with accessible porosity from a meltable cobalt imidazolate framework"
L. Frentzel-Beyme, M. Kloß, R. Pallach, S. Salamon, H. Moldenhauer, J. Landers, H. Wende, J. Debus, S. Henke*, J. Mater. Chem. A, 2018, DOI: 10.1039/C8TA08016J.
MOF glasses represent a new class of functional materials which might have a number of advantages against their crystalline counterparts. We have developed the very first cobalt-based zeolitic imidazolate framework (ZIF) that can be melted and transformed into a glass. In collaboration with colleagues from the Physics Departments of TU Dortmund and the University of Duisburg-Essen, we investigated the structural, thermodynamic and magnetic properties of this new material. Importantly, the liquid and glass phases of the ZIF preserve almost 50% of the porosity of the crystalline parent material. This finding might pave the way for the application of liquid and glassy MOFs in gas separation processes and catalysis.
MOF 2018 in Auckland, New Zealand

Louis, Roman und Sebastian presented the freshest results from the group's research at the 6th International Conference on Metal-Organic Frameworks & Open Framework Compounds ‘MOF 2018’ in Auckland, New Zealand. It has been a fantastic conference with lots of fascinating science, excellent talks and great people. We are looking forward to “EuroMOF 2019" in Paris next year and ‘MOF 2020’ in Dresden in two years.
DAAD-Travel Grant for Louis Frentzel-Beyme

We are delighted that Louis received a travel grant from the German Academic Exchange Service (DAAD) to present his work on porous sodium-organic frameworks at the 6th International Conference on Metal-Organic Frameworks & Open Framework Compounds ‘MOF2018’ in Auckland, New Zealand, this year. Great job!.
Funding for Porous Salts

Most MOFs are based on di-, tri- or tetravalent metal ions (e.g. Zn2+, Cu2+, Al3+, Zr4+ etc.). Porous frameworks composed of monovalent alkali ions (Li+, Na+, K+) linked by organic anions are rare, however. We are very happy that the DFG decided to fund our project on “Porous Alkali-Organic Frameworks - From Design towards Application”. First examples of these new materials, which can be regarded as porous alkali-organic salts (see Figure), will be reported soon.
Max-Buchner-Scholarship

Sebastian received a Max-Buchner-Scholarship from DECHEMA for a research project focussing on the utilisation of nanoparticles of flexible MOFs as functional additives for lubrication systems.
EXMAC

We are part of the EXPLORE Materials Chain (EXMAC) project, which enables us to invite an international postdoc to our lab for two weeks (27 October – 14 November 2018).
Within this two-week stay, we will develop a joint research idea and prepare a dedicated proposal for the independent funding of the postdoc. If you are interested to visit our group and work on an exciting project of current materials chemistry please visit our profile on the EXMAC webpage.
Top Download
Our recent paper “Different Breathing Mechanisms in Flexible Pillared-Layered Metal-Organic Frameworks − Impact of the Metal Center” is among the Top 20 most downloaded articles of Chemistry of Materials in March 2018.
Paper published in Chemistry of Materials
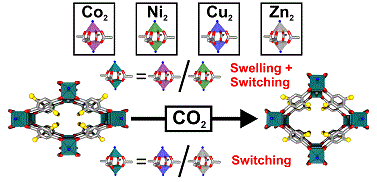
A. Schneemann, P. Vervoorts, I. Hante, M. Tu, S. Wannapaiboon, C. Sternemann, M. Paulus, D. C. F. Wieland, S. Henke*, R. A. Fischer*, Chem. Mater. 2018, DOI: 10.1021/acs.chemmater.7b05052
Flexible metal-organic frameworks expand their extended network structure upon adsorption of gases. We reveal that the mechanism of structure expansion (the so called breathing) can be very different even in isostructural compounds possessing varying divalent metal ions M2+ (i.e. Co2+, Ni2+, Cu2+ or Zn2+). With the help of isothermal gas adsorption measurements and synchrotron X-ray diffraction studies, we revealed that flexible pillared-layered MOFs either switch between discrete phases (M2+ = Cu2+ or Zn2+) or undergo a continuous swelling followed by discontinuous switching (M2+ = Co2+ or Ni2+) upon adsorption of CO2 from the gas phase.
Paper published in Chemical Science
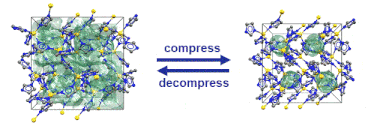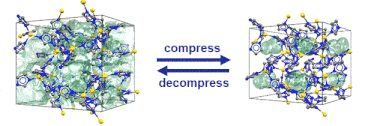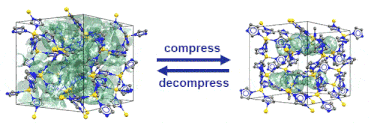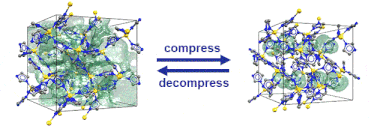
“Pore closure in zeolitic imidazolate frameworks under mechanical pressure”
S. Henke*, M. T. Wharmby, G. Kieslich, I. Hante, A. Schneemann, Y. Wu, D. Daisenberger, A. K. Cheetham, Chem. Sci. 2018,9, 1654-1660
In collaboration with colleagues from Diamond Light Source, Cambridge, Munich and Bochum we discovered that zeolitic imidazolate frameworks of the cag topology reversibly switch between an open and a closed pore form in response to mechanical pressure.